Introduction: CFI-400945 is a first-in-class, potent, selective, orally active inhibitor of Polo-like kinase 4 (PLK4) (Ki=0.26nM), a master regulator of centriole duplication, necessary for genomic integrity (Mason et al. Cancer Cell 2014; 26:163-76). CFI-400945 has activity in leukemia cell lines and primary leukemia samples including those with complex karyotype, inversion 3 and monosomy 7 (Minden. personal communications). This suggests that CFI-400945 may provide an effective treatment of patients with AML. The objectives of this phase 1 trial was to establish the safety, tolerability, and recommend phase II dose (RP2D) of CFI-400945 in patients with acute myeloid leukemia (AML) and myelodysplastic syndrome (MDS).
Methods: Patients with relapsed/refractory AML or MDS and patients with untreated AML who refused induction chemotherapy or who are not candidates for intensive chemotherapy were eligible. Dose escalation followed a standard 3+3 design with a starting dose of 64 mg orally once daily. Plasma levels of CFI-400945 free base were measured on Days 1, 2, & 29 of Cycle 1 and Day 15 on all subsequent cycles. Peripheral blood and/or bone marrow were obtained at baseline, Day 8 of Cycle 1 and Day 1 of each subsequent cycle prior to dosing for pharmacodynamic monitoring. Safety assessments using the NCI CTCAE version 4.03 were performed.
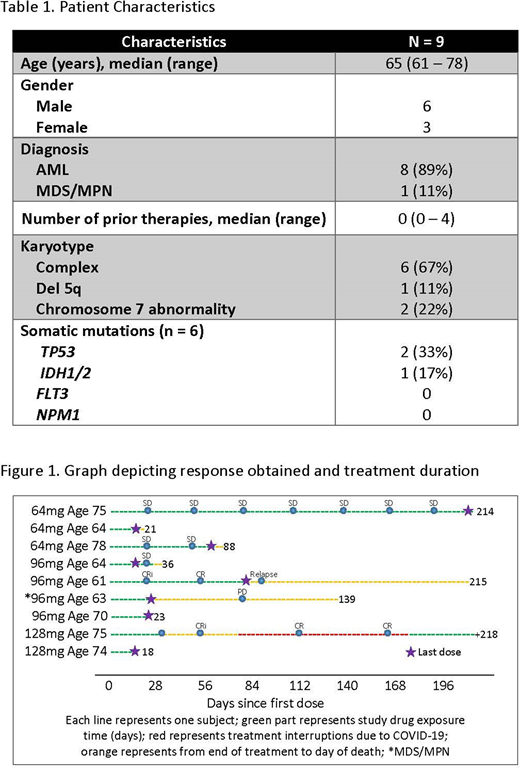
Results: From May 2018 to June 2019, nine patients have been enrolled on study across three pre-defined dose levels (64 mg [n=3], 96 mg [n=4], and 128 mg [n=2]). Three patients had untreated AML, five patients had relapsed/refractory AML and one patient had myelodysplastic syndrome/myeloproliferative disorder (MDS/MPN). Patient characteristics at diagnosis are outlined in Table 1. Six (67%) patients had baseline high throughput sequencing; the most frequent mutations were TP53 (33%), TET2 (33%), KRAS (33%) and DNMT3A (33%). A total of 20 cycles were administered with a median of 1 cycle (range, 0 to 7 cycles).
The most common non-hematological drug related toxicities of any grade, which occurred in over 20%, were diarrhea (44%), headache (44%), colitis (33%), vomiting (33%), bilirubin increase (22%), dizziness (22%), fatigue (22%), and nausea (22%). One patient on the 96 mg dose level was not evaluable for DLT and hence, replaced. Both patients treated at the 128 mg/day dose level developed DLTs, consisting of grade 3 colitis and grade 5 sepsis and colitis. Pharmacokinetic profile indicated low interpatient variability between patients. Maximum exposure did not correlate with toxicity
Six patients were evaluable for disease response. Two (33%) achieved complete remission (CR), 3 pts (50%) had stable disease (with one patient having a 78% reduction in marrow blast count). The patient with MDS/MPN who did not complete 1 cycle of therapy progressed to AML (Figure 1). Both patients who obtained a CR had an early response within 2 cycles. One CR has been durable for 218 days with no measurable residual disease (MRD) by flow cytometry. The additional patient, who obtained a CR with incomplete platelets recovery, with subsequent best response of CR, had a sustained response for 91 days before relapse was confirmed by bone marrow examination (Figure 1).
Conclusion: Single agent CFI-400945 has activity in patients with poor risk AML. The RP2D in this population is 96 mg once daily. Dose expansion is occurring at the RP2D level. A phase 2 study with CFI-400945 single agent or in combination study with azacitidine or decitabine is planned.
Leber:Novartis: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Abbvie: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Alexion: Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Lundbeck: Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Takeda/Palladin: Honoraria, Membership on an entity's Board of Directors or advisory committees; Treadwell: Honoraria, Membership on an entity's Board of Directors or advisory committees; Amgen: Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; BMS/Celgene: Honoraria, Membership on an entity's Board of Directors or advisory committees; Pfizer: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Otsuka Pharmaceutical: Honoraria, Membership on an entity's Board of Directors or advisory committees; Janssen: Honoraria, Membership on an entity's Board of Directors or advisory committees. Bray:Treadwell Therapeutics: Current Employment; TIO Discovery: Current Employment. Gupta:Pfizer: Consultancy; Novartis: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Sierra Oncology: Consultancy, Membership on an entity's Board of Directors or advisory committees; Bristol MyersSquibb: Honoraria, Membership on an entity's Board of Directors or advisory committees; Incyte: Honoraria, Research Funding. Maze:Novartis: Honoraria; Pfizer: Consultancy; Takeda: Research Funding. McNamara:Novartis: Honoraria. Schimmer:Jazz: Honoraria; Otsuka: Honoraria; Medivir AB: Research Funding; AbbVie Pharmaceuticals: Other: owns stock ; Takeda: Honoraria, Research Funding; Novartis: Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal